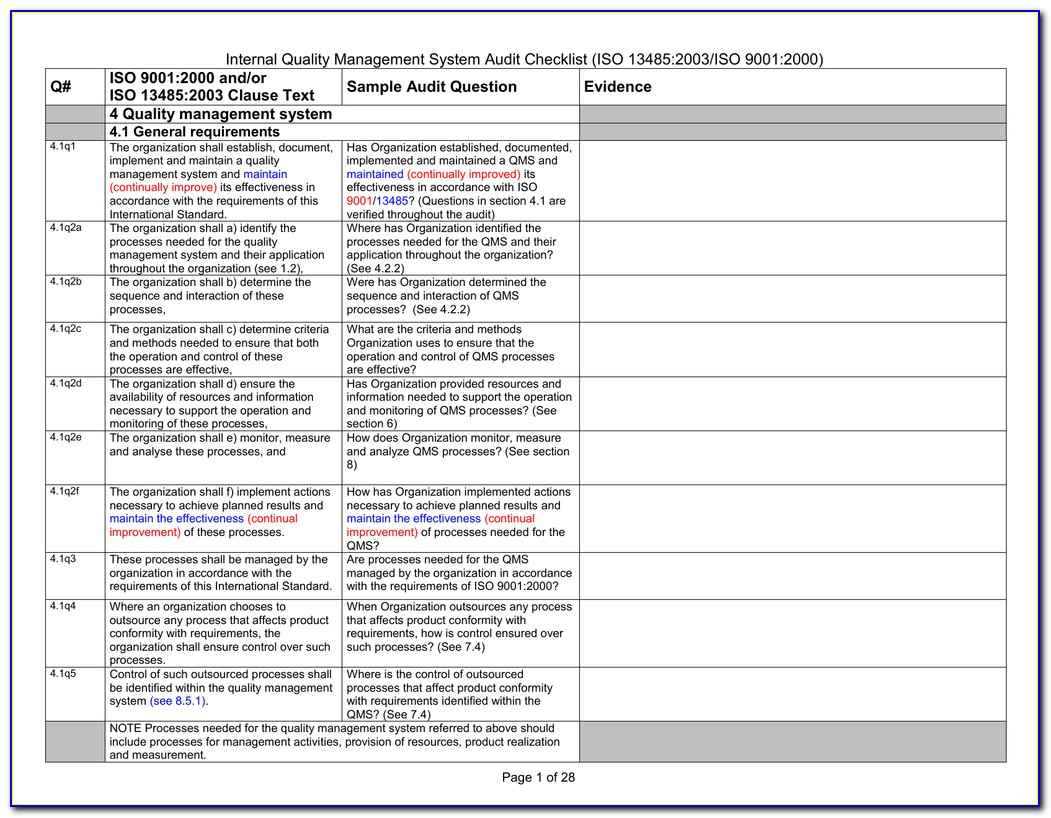
These include:Ībility to meet requirements – How well does the supplier meet your requirements? Can they meet the tolerances you need? Are they compliant with any legal requirements that are necessary to provide their products? For example, if there is a certificate of quality or certificate of analysis for purchased material, are the tolerances listed on the certificate in accordance with your requirements? So, how do you make sure that suppliers will meet your needs? ISO 13485 contains a list of things to consider when approving a supplier. This list, which includes the dates for re-evaluations, ensures that you always use a supplier who is in good standing with your company.Ĭriteria for evaluation and selection of suppliers While not strictly a requirement of the standard, many companies maintain a list of the suppliers who are currently approved for use by the purchasing department. 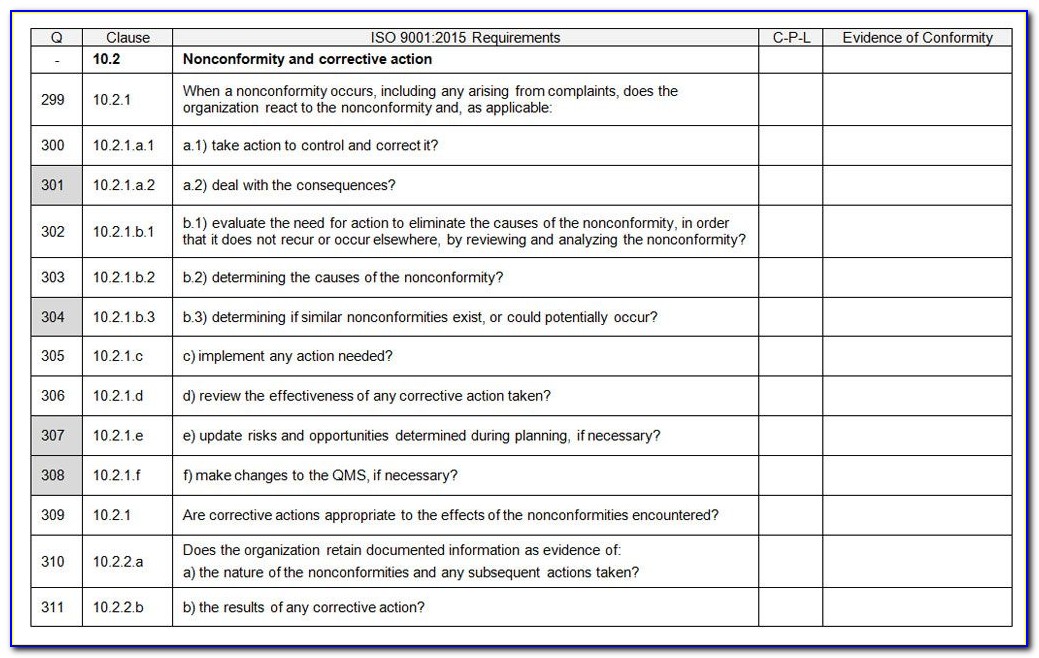
This approval, monitoring, and any necessary correction of suppliers needs to be demonstrated by records that are maintained to demonstrate that the suppliers are capable of meeting requirements. Failure to meet requirements needs to be addressed and corrected as soon as it is noticed. Supplier evaluation is not intended to be a one-time thing: once a supplier is approved for use, monitoring of performance is crucial to ensure that they continue to meet your requirements. Your supplier evaluation, and subsequent supplier monitoring, are intended to be based on the risks that each supplier poses to your ability to provide medical devices that meet customer and regulatory requirements. One of the purchasing controls to ensure that you get the right products for your medical device manufacturing is supplier evaluation. ISO 13485:2016 supplier management requirements Risk to the medical device – Along with quality, how does the supplied product affect the safety of the medical device?.Effect on quality – How much of an effect does the supplied product have on your medical device manufacturing?.Supplier performance – How well does the supplier meet your needs?.Ability to meet requirements – How well does the supplier meet your requirements?.For any clause that is determined to be not applicable, the organization records the justification as described in 4.2.2. If any requirement in Clauses 6, 7 or 8 of ISO 13485:2016 is not applicable due to the activities undertaken by the organization or the nature of the medical device for which the quality management system is applied, the organization does not need to include such a requirement in its quality management system. It is the responsibility of the organization to ensure that claims of conformity to ISO 13485:2016 reflect any exclusion of design and development controls. These regulatory requirements can provide alternative approaches that are to be addressed in the quality management system. 
If applicable regulatory requirements permit exclusions of design and development controls, this can be used as a justification for their exclusion from the quality management system. The processes required by ISO 13485:2016 that are applicable to the organization, but are not performed by the organization, are the responsibility of the organization and are accounted for in the organization's quality management system by monitoring, maintaining, and controlling the processes. Wherever requirements are specified as applying to medical devices, the requirements apply equally to associated services as supplied by the organization. Requirements of ISO 13485:2016 are applicable to organizations regardless of their size and regardless of their type except where explicitly stated. ISO 13485:2016 can also be used by suppliers or external parties that provide product, including quality management system-related services to such organizations. Such organizations can be involved in one or more stages of the life-cycle, including design and development, production, storage and distribution, installation, or servicing of a medical device and design and development or provision of associated activities (e.g. ISO 13485:2016 specifies requirements for a quality management system where an organization needs to demonstrate its ability to provide medical devices and related services that consistently meet customer and applicable regulatory requirements.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
